World Get Its First Malaria Vaccine: How it Works & Who Should Take It
Mosquirix, the malaria vaccine by GlaxoSmithKline has already been administered to over 8,00,000 children in 2.3 million doses.
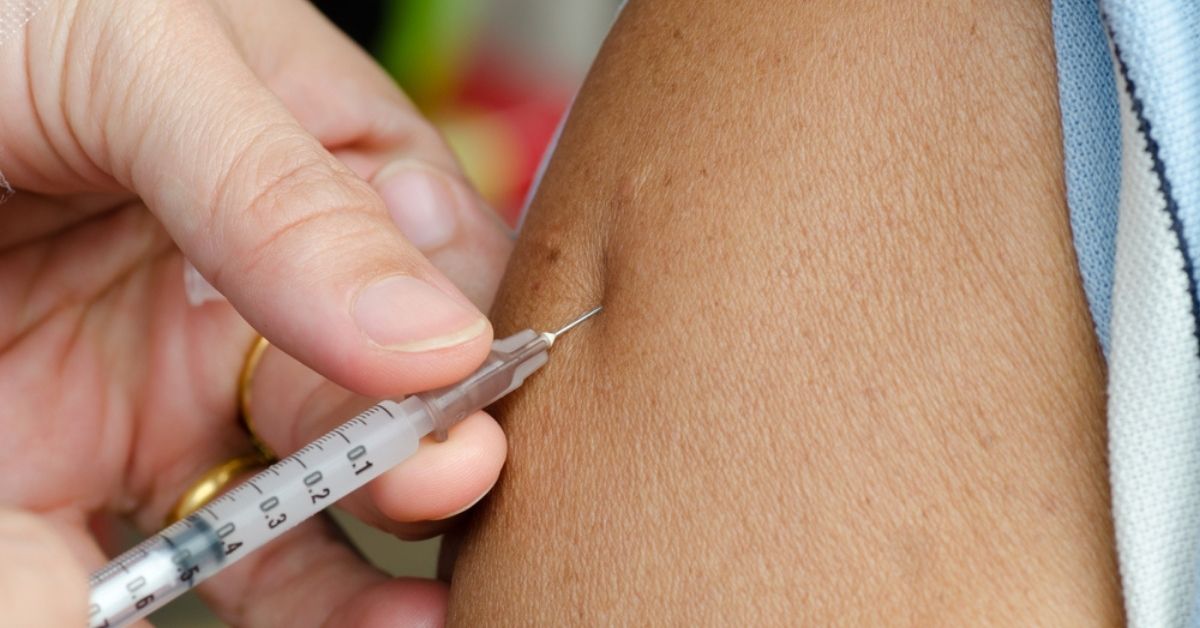
The quest to find a vaccine to combat malaria has been underway for over a century.
The disease kills about half a million people each year, nearly all of them in sub-Saharan Africa, including 2,60,000 children under the age of five. While the number of malaria deaths in India has seen a steady decline over the years, India still accounted for 88 per cent of malaria cases and 86 per cent of malaria deaths in the WHO South-East Asia Region in 2019.
A new vaccine, made by GlaxoSmithKline, boosts a child’s immune system to thwart Plasmodium falciparum, the deadliest of five malaria pathogens and the most prevalent in Africa. The World Health Organisation (WHO) has endorsed the vaccine, which is the first step in a process that should lead to wide distribution in other countries. Dr Pedro Alonso, Director of the WHO’s global malaria program, said, “To have a malaria vaccine that is safe, moderately effective and ready for distribution is a historic event.”
Malaria is a life-threatening disease caused by parasites that are transmitted to people through the bites of infected female Anopheles mosquitoes.

The illness starts with symptoms such as fever, headaches and chills and if it remains untreated, can progress quickly to severe illnesses or death. It is, however, preventable and curable. Children aged under five years of age are the most vulnerable group affected by malaria.
What we know so far:
- Malaria is a rare disease in developed countries and continues to be endemic in developing countries, especially in sub-Saharan Africa and India.
- Mosquirix (RTS,S/ASO1 (RTS.S), the vaccine that has been developed, is not just a first to fight malaria but also the first to be developed to fight a parasitic disease.
- According to a report by the Indian Express, this is the first malaria vaccine that has completed the clinical development process, and received a positive scientific opinion from the European Medicines Agency (EMA).
- The structure of parasites are more difficult to fight when compared to bacteria and viruses.
- In clinical trials, the vaccine had an efficacy of about 50 per cent against severe malaria in the first year, but the figure dropped close to zero by the fourth year.
- A recent trial of the vaccine in combination with preventive drugs given to children during high-transmission seasons found that the dual approach was much more effective at preventing severe disease, hospitalisation and death than either method alone.
- In some cases, children have contracted malaria several times over the years and this alters their immune system leaving them weak and prone to other pathogens.
- Mosquirix is given in three doses between ages five months to 17 months, and a fourth dose roughly 18 months later.
- Post clinical trials, the vaccine was administered to children in Kenya, Malawi and Ghana.
- So far more than 2.3 million doses have been administered to over 8,00,000 children so far.
While the WHO has recommended the widespread use of the first-ever vaccine for malaria, more details on when the vaccine is likely to be available in India are awaited.
(Edited by Yoshita Rao)
This story made me
- 97
- 121
- 89
- 167
Tell Us More
We bring stories straight from the heart of India, to inspire millions and create a wave of impact. Our positive movement is growing bigger everyday, and we would love for you to join it.
Please contribute whatever you can, every little penny helps our team in bringing you more stories that support dreams and spread hope.


















